Drug detection with high-sensitivity using ToF SIMS
The high attrition rate of pharmaceutical drug compounds adds enormously to the cost of those that make it to market, so there is an urgent and growing need to identify failure at earlier stages of drug development.
In order to do so, researchers require as much information as possible. Specifically, there is a need to measure the concentration of a drug at the target in order to accurately predict its pharmacological effect. This then requires a means of generating label-free sub-cellular imaging, as fluorescent labels may affect drug chemistry, altering results.
Time of flight secondary ion mass spectrometry (ToF SIMS) is a powerful tool for label-free chemical imaging, having typically very high lateral resolution capable of resolving sub-cellular features with 3D analysis capabilities.
ToF SIMS is thus a potentially powerful analysis tool for the screening of new drug compounds. However, the use of high energy projectiles for ToF SIMS analysis can cause molecules to fragment, preventing the molecular ion from being detected. This can lead to a lot of ambiguity, for example distinguishing between a drug compound and its metabolites.
Another possible stumbling block is the issue of sensitivity, particularly for those compounds of most interest. In a recent study by the National Physical Laboratory (NPL), Vorng et al. demonstrate that the sensitivity in ToF SIMS is proportional to the Log P of that compound, such that compounds with low or negative Log P values are extremely difficult to detect.
Log P, or partition coefficient, is a measure of hydrophobicity, and is a major factor used in pre-clinical assessment of a compound’s druglikeness. It is advisable that a drug candidate be as hydrophilic as possible while still retaining adequate binding affinity to the therapeutic protein target, i.e. that the Log P be as low (or negative) as practicable. This presents an obvious problem for the use of ToF SIMS as an analytical tool in this context.
We have recently led the development of a new type of ion source for ToF SIMS that provides unparalleled sensitivity particularly for organic species. Available exclusively on the J105 SIMS, the Water Cluster Source simultaneously reduces fragmentation while increasing ionization, for truly unparalleled sensitivity of drugs, metabolites, biomarkers, lipids, peptides and more.
Combining this new ion source with the already impressive sensitivity of the J105 SIMS, even low Log P compounds can be detected in tissue and cells, with direct, label-free imaging of the molecular compounds at sub-cellular resolutions.
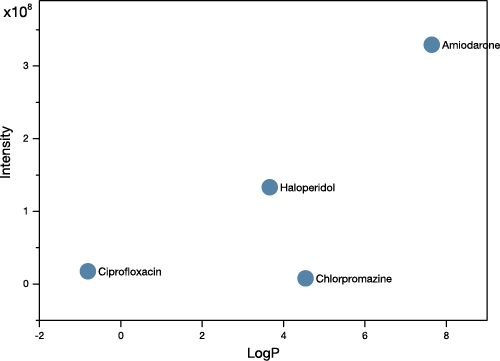
To demonstrate this, we doped tissue homogenate with 4 different pharmaceutical compounds that span the range of Log P from -0.8 to 7.6. The relationship between sensitivity and Log P reported by NPL is observed in this data, however the slope of the line is greatly reduced, with only a factor of 40 between the highest and lowest values.
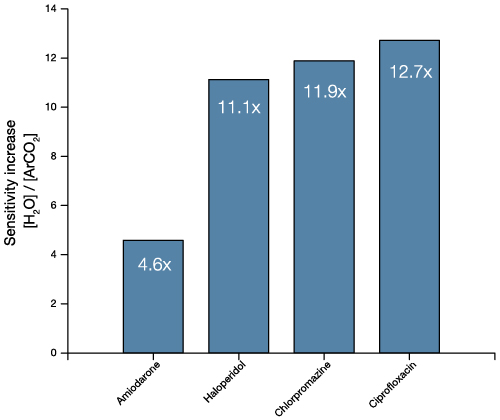
As a comparison, we performed the same experiments with a state-of-the-art Ar gas cluster ion beam and plotted the yield against that of the new Water Source. The Water Source increased sensitivity by an order of magnitude in most cases, with the largest increase being for those compounds with the lowest Log P values. This indicates that the improvement in sensitivity is greatest for those compounds that need it the most.
As a final demonstration of the capabilities of the J105 with the Water Source, we performed tandem MS analysis on the homogenate samples. Tandem MS is an important step for confirming any assignment in mass spectrometry, however the inefficiency of the process often means it can only be performed on high intensity peaks. With the boost in sensitivity provided by the Water Source, tandem MS analysis is possible even on compounds with relatively low Log P values, such as ciprofloxacin.
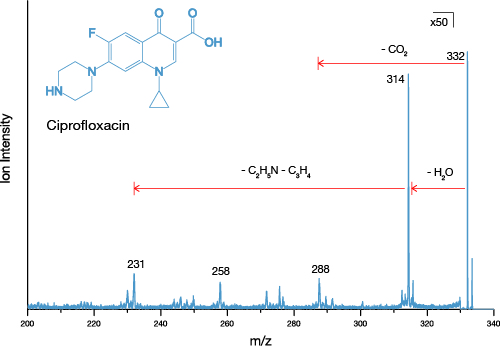
ToF SIMS is a potentially powerful analysis tool for the screening of new drug compounds, however research is hampered by the inherently low sensitivity to many drug candidates. The J105 SIMS in combination with the Water Cluster Source provides unparalleled sensitivity to drug compounds, particularly in complex matrices such as tissue and cells, even for low Log P compounds. This unprecedented sensitivity combined with sub-cellular imaging and high-resolution 3D imaging mean the J105 SIMS is a powerful tool for drug analysis.
To learn more about how the J105 SIMS can benefit your research or to set up a demonstration, get in touch via our Contact Page.

 Ionoptika 2020
Ionoptika 2020 
 Ionoptika
Ionoptika  Ionoptika Ltd
Ionoptika Ltd  Ionoptika Ltd
Ionoptika Ltd